 Nitrogen and oxygen will not condense at these temperatures since their intermolecular forces are much weaker. There is plenty of actual physical space to go around but once a certain limit is crossed (the dew point) the water will become liquid. 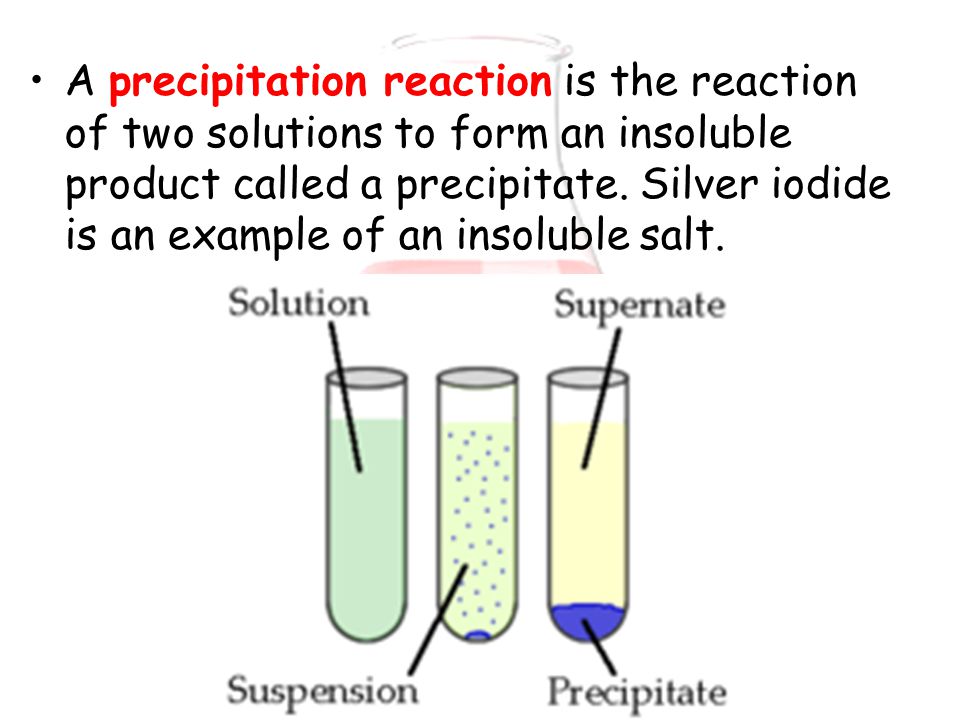
You may compare precipitation to condensation of water in air. Here you might also discover yourself that smaller particles dissolve more (not only faster!) than bigger particles. In summary, the amount of 'space' with respect to solubility is a function of the strength of intermolecular forces and the amount of solvent, among other things. If the ratio of the amount of solvent to the amount of solute increases, that is we add more solvent, we should expect the solubility to increase. There is often some energy barrier to this approaching, as colloid chemists will tell you, but if the end result is some energy minimum, the process will occur at some rate. Hence, at a certain radii the intermolecular forces become strong enough to pull the solutes together. Instead as you increase the concentration of your solute, the distances between solute entities starts to decrease on average. What does this 'fitting' mean exactly? It definitely does not mean there is not enough physical space per se. 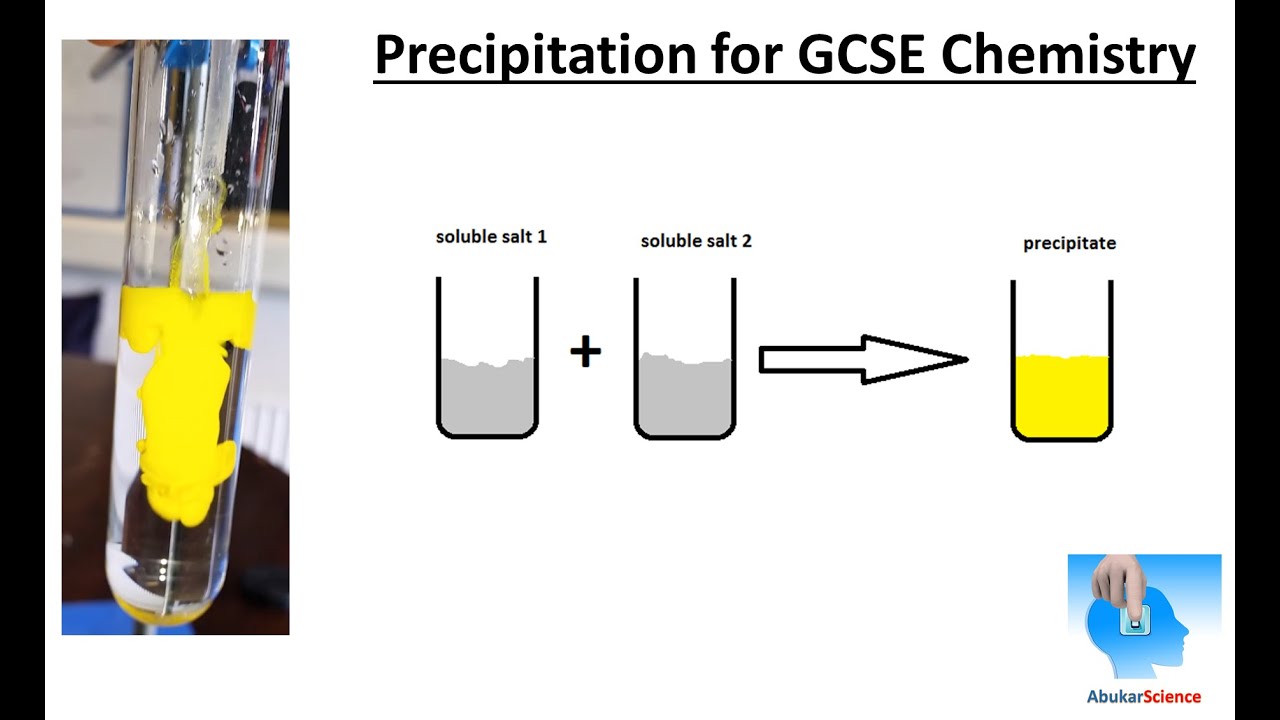
Similar things are true, again in a certain interpretation, for more realistic situations. Further, chemical reactions consist of chemical changes that take place within the substances. 
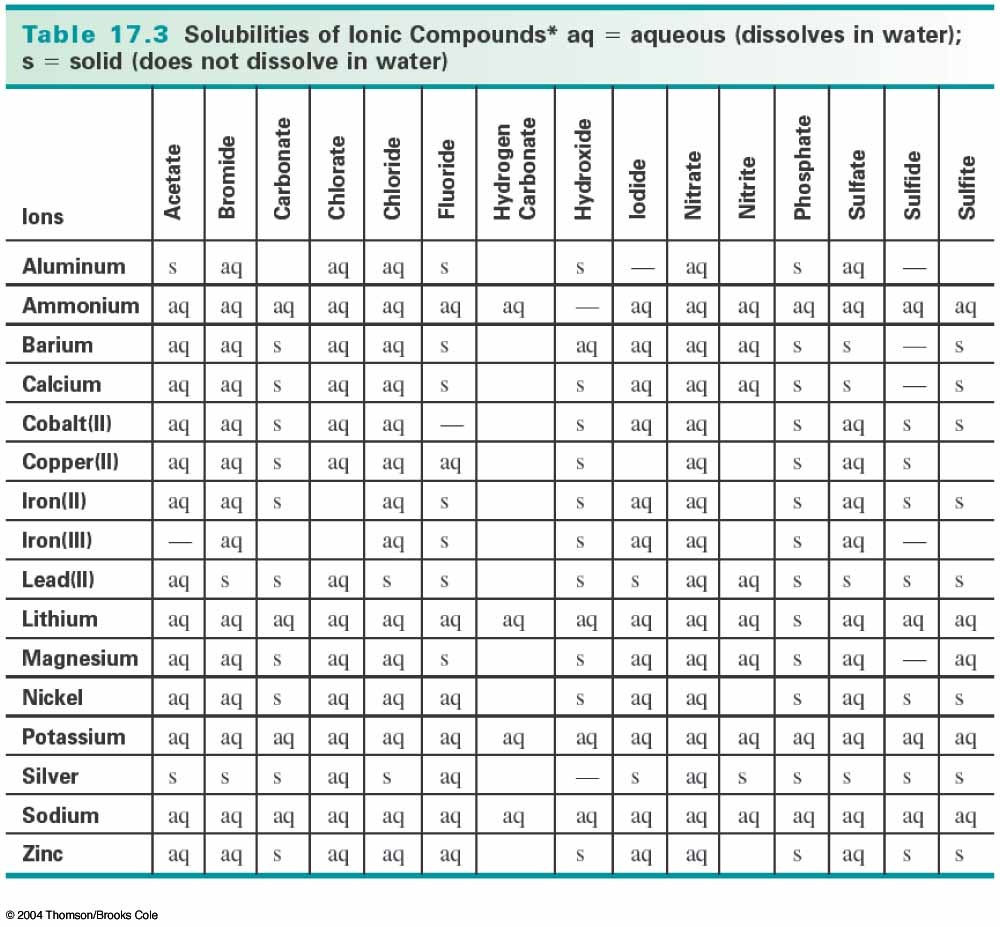
In some sense, the solid 'does not fit' into the liquid droplet over a certain limit. A precipitation reaction is a chemical reaction that occurs in aqueous solution and form precipitates. 1 a : to throw violently : hurl the quandaries into which the release of nuclear energy has precipitated mankind A. As an adjective precipitate is headlong falling steeply or vertically. Yes, there will be some adsorption and solubility here, yet most of the solid will remain undisturbed. chemical precipitation, formation of a separable solid substance from a solution, either by converting the substance into an insoluble form or by changing the composition of the solvent to diminish the solubility of the substance in it. precipitate deposit In transitive terms the difference between precipitate and deposit is that precipitate is to cause (water in the air) to condense or fall to the ground while deposit is to put money or funds into an account. We would not expect the entire solid to disappear into the liquid. Say you have a large hydrophilic solid, and a small drop of water is added on top.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
